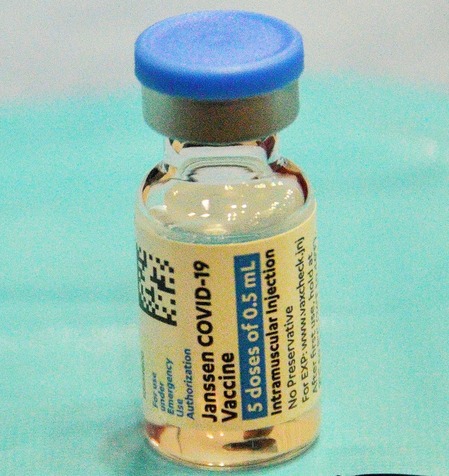
Johnson & Johnson is seeking to conduct a local clinical trial in India for its single-dose COVID-19 vaccine under the new fast-track rules rolled out by the government last week.
The company said on Tuesday it had submitted an application to the Drugs Controller General of India requesting approval for the "bridging" safety and immunogenicity study for its vaccine.
Last week, the government had eased rules to fast-track emergency approvals for foreign-made COVID-19 vaccines. The move exempts companies from carrying out long-drawn "bridging" trials for their vaccines which could stretch over several months.
According to the new rules Vaccines for Covid-19, which have been developed and are being manufactured in foreign countries and which have been granted emergency approval for restricted use the US, UK, Europe and Japan or which are listed in WHO (Emergency Use Listing) may be granted emergency use approval in India.
The new rules state that the first 100 beneficiaries of a foreign vaccine shall be assessed for seven days for safety outcomes before it is rolled out for further immunisation programme within the country.
J&J will be eligible to import the vaccine once it gets this clearance.
J&J also has a deal with Indian firm Biological E Ltd to contract-manufacture its vaccine in India.
Besides, Johnson & Johnson has the support of the Indo-Pacific Quad agreement between the US, India, Japan and Australia to expedite vaccine production with an Indian partner as most countries do not enough doses to fight the deadly Covid-19. Quad agreement signed after Joe Biden took over as US President provides for US and Japanese finance coming together with Indian manufacturing capacity to roll out more vaccines for the war against the pandemic.